Research Areas
In the laboratory, we are deeply passionate about many different aspects of how host myeloid cells respond to and are shaped by inflammation. While our major focus is in pancreatic and lung cancer, we are extremely interested in defining shared myeloid cell biology across disease contexts (e.g.– sepsis, fibrosis, acute radiation damage).
“How does tumor-associated myelopoiesis impact BM cells epigenetically?”

Our prior work has started revealing the impact of cancer inflammation on chromatin state of myeloid progenitors. We are now interested in understanding the mechanistic details of how tumor cues alter chromatin and/or DNA methylation at consequential gene loci, and how long-lasting these changes are. We are developing cutting-edge assays to integrate knowledge from transcriptome, epigenome, DNA architecture, with a focus on molecular pathways involved in oxidative stress regulation, RNA quality control, and proteostasis.
“What are the metabolic consequences of oxidative stress responses during tumor-associated myelopoiesis?”
Some of the most dominant pathways altered in BM cells by distal tumors involve proteostasis (quality control of protein synthesis/degradation) and oxidative stress. We are particularly keen to study how metabolic maladaptations in myeloid progenitors impacts gene regulation, hyperproliferation, fate changes, and spatial mislocalization. We will use low-input metabolomics approaches with a focus on ER/mitochondrial biology to dissect tumor-inflammation driven metabolic changes in BM and tissue sites.
“Is the steady-state fate of BM stem cells and multipotent progenitors altered by cancer inflammation?”
Chronic inflammation can rewire bone marrow hierarchy, prompting HSCs and multipotent progenitors to undergo rapid, aberrant fate transitions and/or lineage bypassing to meet the urgent demand for myeloid cells, often at the expense of long-term stem cell health and lymphoid diversity. We hypothesize that mitogen-driven emergency myelopoiesis involves the remodeling of constitutive heterochromatin, leading to a more permissive epigenetic landscape. This loss of ‘structure’ can lower the activation barrier for priming of non-canonical gene programs, leading to lineage plasticity within the progenitor compartment. We will exploit detailed molecular profiling, a newer class of lineage tracing, and functional validation to study how cancer inflammation distorts cellular fates.
“How do acute or chronic genotoxic therapies impact reactive myelopoiesis?”
Genotoxic therapies such as chemotherapy and systemic radiation can impose severe physiological strain on the bone marrow in patients, and oxidative stress regulators such as NRF2 emerge as the primary mediators of hematopoietic recovery and myeloid ‘rebound’. Leveraging mouse models and primary human samples, our lab is extremely interested in understanding the short-term (immunosuppression or immunoparalysis) and long-term consequences (innate memory) of such therapies on BM progenitor biology and BM niche architecture, with implications for subsequent susceptibility to secondary cancers or infections.
“Does myelopoiesis in response to therapies influence metastatic dissemination and/or dormancy exit?”
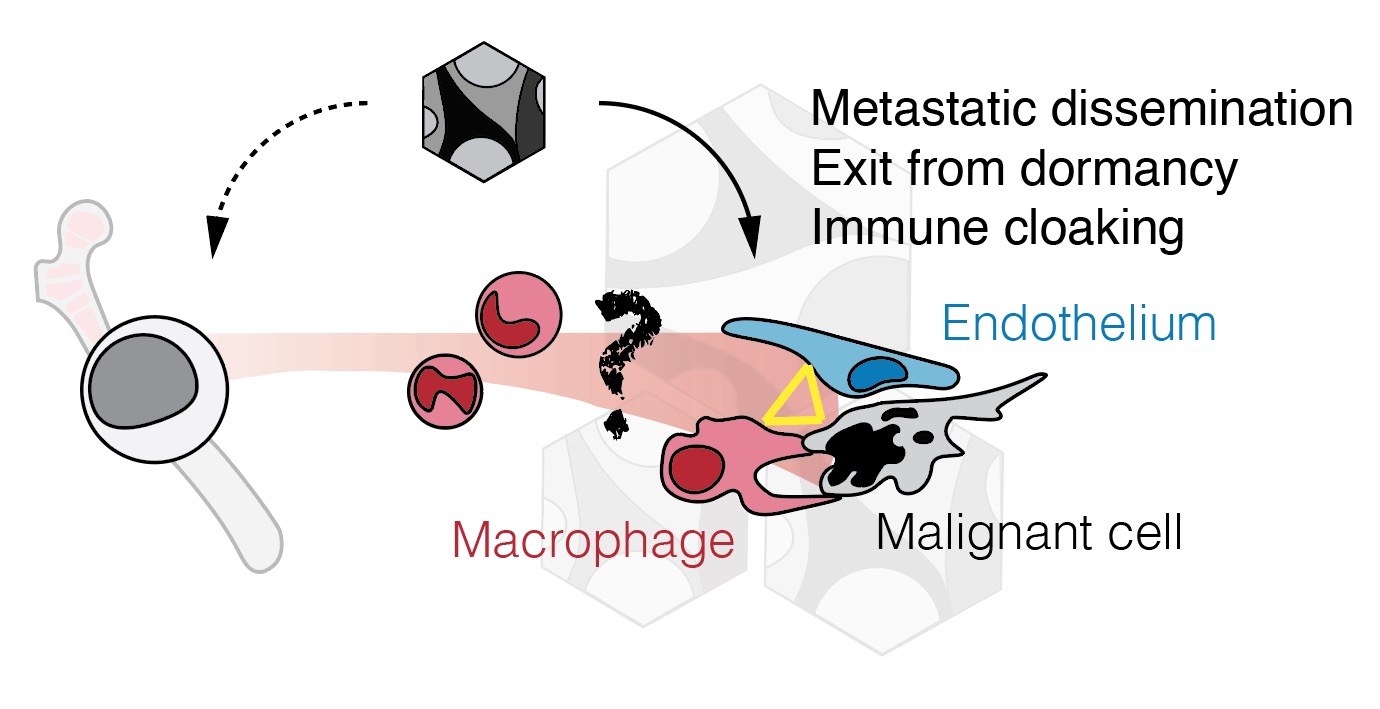
Post-therapy myelopoiesis is important to renewing the host immune system but can also inadvertently trigger the awakening of dormant cancer cells or promote metastatic seeding. There is clear clinical evidence that chemotherapies can result in a myeloid ‘reactivity’, releasing a surge of immature immunosuppressive monocytic/neutrophilic cell states (often termed MDSCs). We are very excited to be working within the larger Cancer Dormancy Institute ecosystem to address the systemic interplay between myeloid rebound and subsequent metastatic/relapse risk. The ultimate goal is to develop myeloid-targeting strategies that complement existing therapies to limit relapse risk while sparing homeostatic function.
We are constantly evolving in our thinking about myeloid immunity, and our project directions reflect that. Get in touch if you want to learn more or participate/collaborate!
Our Funding
- National Institutes of Health (NIH) – National Cancer Institute (NCI)
- Montefiore Einstein Comprehensive Cancer Center
Highlighted
Please visit the Research page for more details on publications